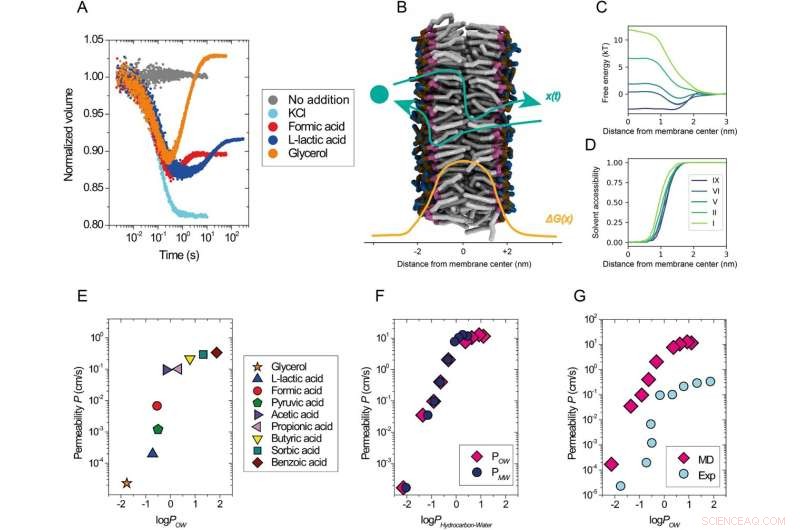
Perméabilité des solutés en fonction de leur lipophilie. Un aperçu du test expérimental. Données cinétiques obtenues avec le test d'auto-extinction de la calcéine à l'aide de vésicules composées de DOPC mélangées à un tampon (gris) ou soumises à un choc osmotiquement avec du KCl 52,5 mM (cyan), du formiate de sodium 50 mM (rouge), du L-lactate de sodium 50 mM (bleu) ou 120 mM de glycérol (orange) à 20 °C. B Description schématique du processus de perméation, x(t), à travers une membrane lipidique avec un exemple de profil d'énergie libre, ΔG(x) (queues lipidiques, gris ; fragment glycérol, violet ; fragment phosphate, ocre ; et fragment choline, bleu ; les molécules d'eau ne sont pas représentées). C Profils d'énergie libre sélectionnés à partir de simulations de solutés avec différents niveaux d'hydrophobicité (I le plus hydrophile, IX le plus hydrophobe) traversant une membrane lipidique DOPC en fonction de la distance du centre de la bicouche le long de la membrane. Seule la moitié de l'ensemble du profil de perméation symétrique est représentée. D Profils d'accessibilité aux solvants des solutés pénétrants le long de la voie de perméation. Les solutés interagissent avec les molécules de solvant même profondément dans la région de la queue de la membrane (x < 1,0 nm). Coefficients de perméabilité E–G pour les membranes DOPC provenant d'expériences à 20 °C (E, G) et de simulations (F, G) tracées par rapport au logarithme de l'octanol/eau (logPOW ) et coefficient de partage membrane/eau (logPMW ) des solutés. Solutés des niveaux hydrophobes I (logPOW = −2.14) à IX (logPOW = 1,1) ont été utilisés dans les simulations. JournalP est log10 (coefficient de partage), où P fait référence à la distribution d'équilibre d'une molécule entre la phase hydrophobe et hydrophile de deux solvants non miscibles. Le log expérimentalPOW les valeurs des acides faibles et du glycérol (E, G) proviennent de la base de données PubChem (https://pubchem.ncbi.nlm.nih.gov/). Le journal simuléPOW les valeurs (F, G) ont été tirées du travail25 ; logPMW les valeurs (F) ont été obtenues à partir des simulations actuelles. Les coefficients de partage sont liés à la différence d'énergie libre entre les phases respectives. Dans le cas de PMW , c'est la différence entre le centre de la membrane et la phase solvant. Les valeurs de l'énergie libre sont présentées en (C). Crédit :Nature Communications (2022). DOI :10.1038/s41467-022-29272-x
Les bactéries, les champignons et les levures excrètent très bien des substances utiles telles que les acides faibles. Une façon dont ils le font est par diffusion passive de molécules à travers la membrane cellulaire. At the same time, cells need to prevent leakage of numerous small molecules. Yeast cells, for instance, can live in hostile environments thanks to a very robust and relatively impermeable membrane system. Biochemists at the University of Groningen have studied how the composition of the membrane affects passive diffusion and the robustness of the cell membrane. Their results, which were published in Nature Communications on March 25, could help the biotech industry to optimize microbial production of useful molecules and help in drug design.
Border control is very important to cells. Their membranes separate the inner and outer environments, which are quite different. To absorb useful compounds, such as nutrients, or to excrete waste, cells can use selective transport systems. However, some transport across the membrane takes place by passive diffusion. This is a non-selective process that will let some molecules go in or out, depending on their size and hydrophobicity, for example. Active transporters have been studied extensively; however, our knowledge of passive diffusion through the membrane is still very incomplete.
Synthetic vesicles
This is a problem for the biotechnology industry, which uses cells as factories to produce a myriad of useful substances and that needs these worker cells to survive under harsh conditions, for example in an environment with a high alcohol or weak acid concentration. Bert Poolman, Professor of Biochemistry at the University of Groningen, was approached by a biotech company that was interested in producing lactic acid in bacteria. They wanted to know more about passive diffusion. This fitted in nicely with another project that Poolman is working on. "We are highly interested in these passive transport processes because of our involvement in a project to build a synthetic cell," says Poolman. "If you can use passive diffusion instead of an active transport system, you need fewer parts to construct such a cell."
So, he combined both questions in a research project. "We started out with a systematic study of what causes the differences in permeability of yeast membranes and bacterial membranes," says Poolman. His team created synthetic vesicles that were made up of three to four different lipids. Ergosterol or cholesterol was added to the membranes to affect their fluidity and rigidity. A range of small molecules was tested using this system and the results from these experiments guided molecular dynamic simulations of diffusion through membranes. The in-silico studies, supervised by Professor Siewert-Jan Marrink, provided a deeper insight into the molecular mechanism of diffusion.
Tweaking
The fatty acid tails of the lipids turned out to be most important in determining the properties of membranes, whereas the hydrophilic head groups had little effect on the permeability. The length of the tails also mattered. "And saturated tails, with no double carbon bonds, are stiffer than unsaturated ones. Hydrophobic interactions cause a close packing of these tails, resulting in a gel phase that is not very penetrable," explains Poolman. Sterols increase the fluidity but in the case of yeast, which uses ergosterol, the permeability remains low. "Thus, by tweaking the saturation of the fatty acids and the type and amount of sterol in the membrane, we can modify the permeability of the plasma membrane of yeast and bacterial cells."
Poolman and his colleagues have, therefore, defined a number of variables that alter the permeability of membranes for different classes of compounds. This information can be used by companies that use yeasts or bacteria as cell factories. "However, our results cannot be directly applied to those cells," says Poolman. "Real membranes contain hundreds of different lipids and the composition can vary between different locations in the membrane. In addition, these cell membranes contain all kinds of proteins. If you make changes in, for example, the lipid composition of the membrane, a lot can go wrong and the function of a membrane protein can be affected."
Drug design
The increased understanding of the physical processes that affect permeability can help companies to understand why certain cells are better for specific processes than others. "The usual way to tweak strains is by directed evolution. Our results will help companies to better understand the results of those optimizations and guide their cell engineering efforts."
Another application is the design of drugs that act inside cells. "Pharmaceutical companies use a set of empirically established rules to optimize drugs for action inside cells, based on parameters such as size or polarity. Our study highlights the importance of the membrane composition of the targeted cells and this could help in drug design."